



Emerging Threats Quarterly Report – Pig Diseases - January-March 2014
This report from the Animal Health Veterinary Laboratories Agency (AHVLA) in Great Britain includes an overview of PED in North America as well as cases in the UK of oedema disease, nervous disease due to sapelovirus, the detection of PCV2b virus and growing diversity of the PRRS virus.Highlights
- Virulent Porcine Epidemic Diarrhoea (PED) virus in North America
- Unusual outbreak of oedema disease causing ataxia
- Nervous disease due to porcine sapelovirus in growers
- Porcine circovirus 2 genotyping detects a PCV2b variant
- Genetic diversity continues to increase in PRRS virus
New and Emerging Diseases
Analysis of diagnostic submissions from which no diagnosis was made
This report reviews VIDA data where a diagnosis was not reached (DNR) despite the sample receiving “reasonable” testing. This allows monitoring of this class with the aim of providing information on potential new or emerging diseases or syndromes. ‘Prior years’ refers to pooled data for 2009-2013 for GB VIDA data.
DNR by Presenting Sign and Syndrome
A total of 21.3 per cent of GB pig submissions in Q1, 2014 did not reach a diagnosis. This was not significantly increased compared to the overall DNR for the same period in prior years of 17.5 per cent. DNR rates for SACCVS (17.5 per cent) and AHVLA (22.3 per cent) were not significantly elevated for this period compared to the same period in prior years.
The DNR for GB submissions with a presenting sign of 'Lame' was significantly increased to 28.6 per cent (2/7) compared to 6 per cent in prior years, however this was based on just two undiagnosed submissions and DNRs for this presenting sign for SACCVS and AHVLA were not significantly increased. The undiagnosed AHVLA submission was of synovial fluid which limits the diagnostic testing which is possible.
The DNR for GB submissions with a presenting sign of “Wasting” was significantly increased to 43 per cent (6/14) from 10 per cent in prior years. Most of the undiagnosed submissions were to AHVLA and the DNR for this presenting sign was significantly increased for AHVLA data to 46 per cent from 8 per cent in prior years. Further details are given below.
Six undiagnosed AHVLA submissions in this quarter with a presenting sign of wasting were reviewed. Two were from the same unit; extensive testing did not reveal an infectious cause and a management problem was considered probable. One was a combination of likely Glässers with enteric disease and rotavirus was detected. In two cases, it is likely that wasting reflected an earlier insult and pigs earlier in the course of disease would be more appropriate diagnostic material. One submission was of faeces only, not allowing full diagnostic testing. Overall, these undiagnosed cases did not suggest a common clinical or pathological condition, or the presence of periweaning failure to thrive syndrome (see http://www.defra.gov.uk/ahvla-en/files/pub-vet-pfts.pdf).
The DNR for GB submissions in 'Systemic and Miscellaneous Syndrome' was significantly increased from 8 per cent in prior years to 15 per cent in Q1, 2014 (11/73). This was due to AHVLA undiagnosed submissions (11/60, 18.3 per cent) and the details of the undiagnosed cases were reviewed. These involved pigs of different ages (two-weeks-old to adult) and with a range of presenting signs ('wasting' to 'found dead'). Three were non-carcass submissions allowing limited investigation. Pathology in some indicated the cause of deaths (pleurisy, pericarditis) but the causative organisms were not isolated, possible due to antimicrobial treatment. They do not suggest an emerging syndrome.
The DNR of 22.9 per cent for GB submissions with a presenting sign of ‘Diarrhoea’ was not significantly higher than the DNR of 19.6 per cent in prior years. For submissions to SACCVS and AHVLA individually, there was no significant change in this DNR (14.3 per cent and 25 per cent, respectively, compared to prior years). For enteric syndrome GB submissions in Q1, 2013, DNR was 24 per cent (AHVLA 26 per cent, SACCVS 19 per cent) which was also not significantly changed from prior years (17 per cent). Although numbers were low and not significant, there was an increase in undiagnosed neonatal and preweaned pig enteric submissions and the details of these were reviewed.
AHVLA submissions with a presenting sign of diarrhoea or in the enteric syndrome which were undiagnosed were reviewed. One of the five submissions was of faeces only and did not allow full diagnostic testing. One submission was of pigs which were not typical of the enteric problem on-farm. Two were live neonatal pig submissions, one of these had villous atrophy suggestive of viral infection, testing for rotavirus and PEDv was negative. On both farms, gilt litters were predominantly affected and disease was not epidemic. Further submissions from DNR cases are encouraged. DNR submissions for diarrhoea as a presenting sign and enteric syndrome are being kept under review in the light of both the emergence and continued spread of porcine epidemic diarrhoea in North America and the reports of a neonatal porcine diarrhoea syndrome in parts of Europe mentioned in previous Emerging Threats reports.
There was no other overall significant increase in DNR for other syndromes or presenting signs for GB, AHVLA or SACCVS data.
Analysis of undiagnosed submissions in Q1, 2014 has not revealed evidence of a new and emerging syndrome in GB pigs.
Virulent Porcine Epidemic Diarrhoea virus in North America
As described in the last quarterly report, porcine epidemic diarrhoea (PED) emerged in 2013 in a virulent form in the United States of America (USA) and spread to Canada in January 2014. By May 2014, the virus was reported from 30 US States and the disease was recently made reportable.
PED PCR remains the test of choice for diagnosis. Experience from the US indicates that it has been more problematic than expected to establish reliable serology and that antibody levels fall quite rapidly after infection. The PED antibody ELISA available in AHVLA uses PED virus isolated in the 1980s (Van Nieuwstadt and Zetstra, 1991) and there is no recent UK PED virus material to evaluate how well the test detects antibody in pigs recovered from current PED virus strains.
Interestingly, initial sequence data obtained from PED PCR-positive faeces from a diagnostic submission to Bury St Edmunds in 2000 has revealed that the sequence from the faecal material is different from the in-vitro adapted isolate which is a variant of CV777. This emphasises the propensity of coronaviruses to change with time and how useful it would be to obtain current virus-infected material.
Batch PCR testing of routine diagnostic samples submitted to AHVLA from pigs with diarrhoea has been undertaken through a combination of BPEX funding and Defra-funded surveillance. This has involved testing 143 samples in diagnostic submissions from diarrhoeic pigs on 68 pig units collected between June 2013 and April 2014 and no PCR-positive samples have been detected. If any PCR-positive cases are detected, a priority will be to obtain the genetic sequence data and sera from the affected cohort of pigs and herd to use in evaluating the PED antibody ELISA.
The fact that no PED virus has been detected so far in these diagnostic samples suggests that PEDV is not currently a significant cause of diarrhoea outbreaks in pigs in England and Wales. Funding for diagnosis of suspected virulent PED outbreaks, should they occur, remains available from the Defra-funded pig scanning surveillance project (ED1200).
Ongoing Investigations
Klebsiella pneumoniae septicaemia – surveillance for disease-associated strain
Outbreaks of septicaemia due to Klebsiella pneumoniae subsp. pneumoniae (Kpp) have been diagnosed in East Anglia each summer since 2011 when the disease emerged as a cause of disease, mainly in pre-weaned pigs on outdoor units. As described in previous reports, the Kpp isolates from all outbreaks were found to have the same sequence type (ST25) which differed from the ST of archived non-outbreak Kpp isolates. To investigate whether the ST25 Kpp is present in pigs on non-outbreak pig farms, opportunistic surveillance was performed on all pig carcass submissions to AHVLA Bury St Edmunds and Thirsk between September 2012 and October 2013. A total of 42 Kpp isolates were obtained from nasal or tonsil swabs and none were ST25. Thus outbreak-associated Kpp ST25 has, to date, not been found in pigs from units which have not had Kpp outbreaks.
Further information on Kpp septicaemia is available on this link: www.defra.gov.uk/ahvla-en/publication/pig-surv reports and awareness will be raised as summer approaches.
Unusual Diagnoses or Presentations
There were a number of unusual diagnoses this quarter; details of these have been included in monthly AHVLA and SAC CVS reports, www.defra.gov.uk/ahvla-en/publication/pig-survreports-monthly/. These will be kept under review to assess whether they justify initiation of emerging disease investigations. These cases will be highlighted at an AHVLA presentation at the May 2014 Pig Veterinary Society conference to raise awareness amongst practitioners.
Unusual outbreak of oedema disease causing ataxia
Oedema disease was diagnosed at Langford as the cause of nervous signs mainly consisting of ataxia, weakness and recumbency as well as malaise and anorexia. The small herd comprised three groups of pigs; 45 recently weaned, 29 weighing around 17kg, and 14 older pigs weighing between 70 and 130kg.
The pigs were bought in from different units at the same time. At least 14 pigs across all three groups died over about three weeks. Post-mortem findings were non-specific with no typical oedema of the eyelids, gastric mucosa or mesocolon visible. Pulmonary oedema and increased peritoneal fluid with scant fibrin stranding was found in some of the six pigs submitted.
Histopathology supported the possibility of oedema disease when a fibrinoid vasculopathy was detected in brain and lymph node and E. coli serotype O139:K82 (strain E4) was isolated in pure culture from the small intestines of subsequent submissions confirming the cause of disease.
This outbreak was unusual in that oedema was not evident before, or at, post-mortem examination, possibly due to antimicrobial treatment, or because some pigs were euthanased and lesions had not fully developed.
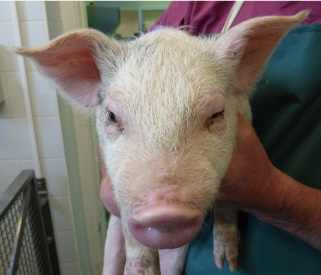
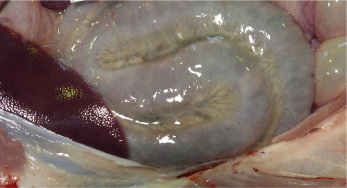
Oedema disease most commonly affects pigs one to four weeks after weaning. Here much older animals were also affected, which may have been due to later challenge following their transport and introduction into an environment contaminated with the oedema disease-producing strain of E. coli. Control can be difficult and centres on hygiene, high quality nutrition, controlling feed levels, and sometimes a reduction in stocking rate. Figure 3 shows the more typical lesions associated with oedema disease.
This case raises awareness of the need to consider oedema disease as a cause of more subtle disease involving nervous signs, even if the characteristic oedema is not evident.
Nervous disease due to porcine sapelovirus in growers
Two outbreaks of nervous disease due to porcine sapelovirus were diagnosed by Bury St Edmunds and Thirsk. These were the first diagnoses of PSV infection by AHVLA since 2008. There was no known connection between the two cases. In both, clinical signs were initially thought to be due to streptococcal meningitis but, when pigs did not respond to treatment with penicillin, this prompted submission of typical cases for post-mortem examination.
In one outbreak there was an upsurge in meningitis-like signs (lateral recumbency and paddling) and sudden deaths in 12-week-old pigs on an all-in, all-out indoor nursery-finisher site. Only one of two sources of 1,000 pigs was affected and around 50 pigs died over a three-week-period, with remaining pigs on the unit growing well.
On the second, pigs on a breeder-finisher unit were affected approximately two weeks after weaning and were reported to be going off their front legs and walking backwards. They progressively worsened, showing nystagmus, then lateral recumbency but no paddling. In both cases, histopathology revealed a non-suppurative polioencephalomyelitis, typical of neurotropic viral infection and immunohistochemistry confirmed involvement of PSV, with no labelling to porcine teschovirus allaying concerns that these were cases of Teschen/Talfan disease. PCR also detected PSV in one of the cases, further confirming the diagnosis.
PSV outbreaks previously investigated by AHVLA have tended to show neurological disease characterised by progressive ataxia and paraparesis, with pigs staying alert. In these outbreaks, clinical signs and pathology were more severe. In one, concurrent PRRS virus infection was detected which may have exacerbated clinical disease. Disease did not persist on the all-in, all-out unit and subsequent batches of pigs from the same breeding source have not, to date, shown similar signs.
On the breeder-finisher unit, successive batches of weaned pigs have been affected over several months and control measures suggested include improved pig flow and hygiene and prompt culling of pigs presenting with signs typical of sapelovirus if they showed no response to antimicrobial treatment. These cases demonstrate the value of investigation of cases not responding to treatment and raise awareness of PSV as a differential diagnosis for meningitis-like signs, along with streptococcal meningitis and oedema disease.
Changes in Disease Patterns and Risk Factors
Porcine circovirus 2 genotyping detects PCV2b variant
Annual VIDA GB diagnoses of PCV2-associated disease (PCVAD) have shown a steady decline as shown below in the last decade largely reflecting widespread use of PCV2 vaccines. Outbreaks diagnosed in the last few years have almost invariably been in unvaccinated pigs or in herds where it was known, or strongly suspected, that groups of pigs had inadvertently not been vaccinated or had some other vaccine administration issue.
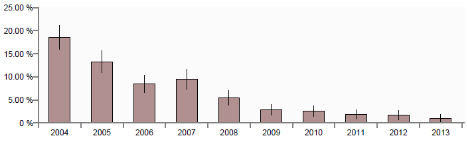
In response to a recent paper on detection of a PCV2 variant in vaccinated pigs with PCVAD (Opriessnig et al, 2013), genetic analysis of the PCV2 virus in 27 cases of PCVAD diagnosed by AHVLA in 2011-13 was undertaken. Nucleic acid was extracted from fixed tissues in which PCVAD was diagnosed and a fragment of PCV-2 genome encompassing the entire coding sequence of open reading frame 2 (ORF2, capsid gene) was amplified and sequenced.
The sequence in 26 cases identified the genotype as PCV2b comprising 23 unique sequences which clustered with representative PCV2b strains from GenBank and with UK PCV2b sequences that were characterised as part of an RVC PMWS project (sampled 2008-2009; unpublished data). The sequence in one case indicates that the virus involved is a PCV2b variant which falls within a distinct cluster which differs from the others sequenced.
Further analysis of the virus in this case is in progress. The outbreak from which this PCV2b variant was sequenced was in pigs which were vaccinated for PRRSv at four-weeks-old and, four weeks later, for PCV2 and Mycoplasma hyopneumoniae and the delay in PCV2 vaccination was suspected to have been significant with respect to PCVAD occurring.
Similar analysis has been initiated on two 2014 cases of PCVAD, in one of which pigs were vaccinated for PCV2 at weaning. PCVAD in most recent cases has involved systemic and respiratory disease with one presenting with the acute pulmonary oedema form of disease as illustrated in Figure 5 below. Analysis of PCV2 in GB pigs prior to 1999 detected genotype PCV2a (Grierson et al, 2004).
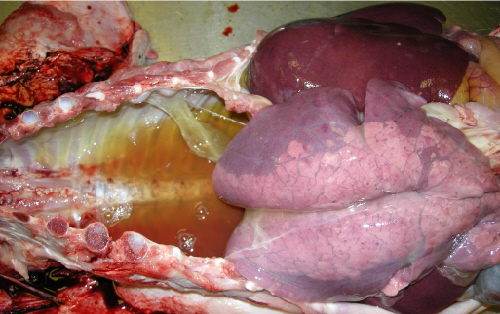
The significance of the detection of the PCV2 variant detected in this case is not yet known, however, within the same cluster, there is a strain which is reported to have been associated with
vaccine breakdown in the US (Opriessnig et al, 2013) and greater virulence in experimental infections (Guo et al, 2012). AHVLA and SACCVS strongly encourage the submission of batches of typically affected pigs to investigate unexplained disease outbreaks. There is useful information on PCV2 genotyping and clinical manifestations of PCVAD on the links below:
- www.pig333.com/circovirosis/importance-and-monitoring-of-the-genetic-makeup-of-pcv2_6793/
- www.pig333.com/circovirosis/update-on-the-diseases-associated-with-the-pcv2-clinic-diagnosis_6440/
Increase in oedema disease incidents GB diagnoses of disease incidents due to Escherichia coli have shown an increased trend in the last three years as shown in Figure 6 with the rate of diagnosis in 2014 (to end March) being equivalent to the rate in 2006.
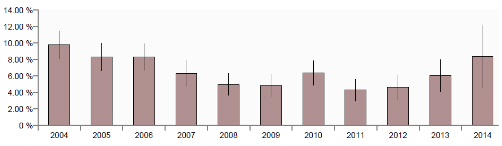
As these incidents include three types of diagnoses, namely enteric colibacillosis, oedema disease or colisepticaemia, these three in the 12 months to end March 2014 were separated as illustrated in Figure 7.
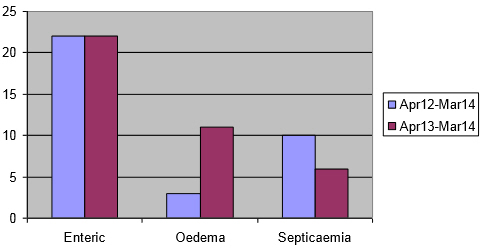
Interestingly, none of the oedema disease diagnoses in the last two years has been in the Bury St Edmunds region. This supports what was suspected from verbal reports from some pig veterinarians and VIOs, that more oedema disease is occurring.
This increase has occurred at the same time as the unusual case described above and other cases which have had subtle lesions. Oedema disease will be highlighted at an AHVLA presentation at the May 2014 Pig Veterinary Society conference to raise awareness amongst practitioners, to help assess if this increase in laboratory diagnoses reflects what is occurring in the field, and to discuss possible reasons for the increase.
Genetic diversity continues to increase in Porcine Reproductive and Respiratory Syndrome virus
The ORF 5 gene sequences were analysed in 82 Porcine Reproductive and Respiratory Syndrome virus (PRRSv) PCR-positive samples from January 2013 to February 2014. These are illustrated in the phylogenetic tree in Figure 8.
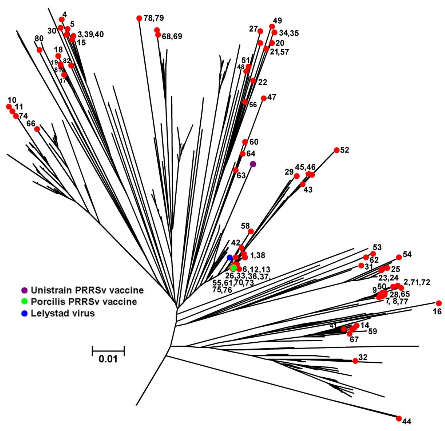
They derive from 59 diagnostic samples and 23 samples from the abattoir baseline survey recently reported. No genotype 2 (North American) PRRSv were detected, providing supportive evidence for the absence of this genotype from the national pig herd.
As anticipated, the most recent sequences obtained show a continuing increase in the diversity of PRRSv strains detected in both diseased and healthy pigs.
The PRRSv virus is prone to a high rate of mutation and recombination and this demonstration of increasing diversity among the population of PRRS viruses confirms previous findings (Frossard et al., 2013). One of the risks of this is that challenges for diagnostic and immunisation methods increase accordingly and work is currently in progress to re-evaluate the performance of the diagnostic PCR.
Amongst the PRRSv strains were two (6 and 70) which were detected in PRRSv-vaccinated pigs with respiratory disease. Immunohistochemistry for PRRSv revealed virus antigen in lung in association with lesions indicating involvement of PRRSv virus in the respiratory disease. The ORF5 sequences of these show 100 per cent homology to the vaccine sequence. Further analysis is in progress to determine the degree of homology in other gene sequences to help determine the relationship of these viruses to vaccine virus and whether they represent recombinants. This illustrates the importance of comprehensive diagnostic investigations and maintaining surveillance of the genetics of PRRSv strains, particularly where clinical problems are persisting or emerge in spite of measures to control disease.
Seasonal increase in PRRS diagnoses Trend analysis of the seasonality of GB PRRS diagnoses shows, as reported in 2012 and 2013, a peak in the rate of diagnoses for the January to March period as illustrated in Figure 9 below, albeit a lower peak than in both the previous years. This is likely to reflect the fact that climatic conditions over the winter months tend to favour survival of the virus and promote transmission.
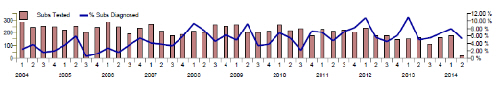
Survival of virus in contaminated pig accommodation, vehicles or on other fomites is also more likely as effective cleaning and disinfection and drying of surfaces is harder to achieve in wet and cold weather. Obtaining an accurate diagnosis in respiratory disease outbreaks assists in determining whether there is viral involvement in disease and allows specific control measures to be implemented. Outbreaks of respiratory disease may involve more than one infectious cause and be combined with systemic disease such as Glassers or PCVAD and these two syndromes (respiratory and systemic) contributed a significant number of diagnostic submissions in January to March 2014 as illustrated in Figure 2.
PRRS, swine influenza and PCV2 viruses detected in these submissions enable ongoing surveillance for early detection of novel or emerging strains which could represent a threat to GB pigs and also form the basis of archived material which is fundamental to future investigations.
Horizon Scanning
Swine deltacoronavirus detected in PED-like outbreaks in North America
In February and March 2014 respectively, the US and Canada confirmed the first detection of a swine deltacoronavirus (SDCV) on pig farms on which there were acute outbreaks of vomiting and diarrhoea but no PED or TGE viruses had been found. The virus has also been found on US farms together with PEDv.
SDCV is clinically similar to, but distinct from, PED and TGE viruses. It is reported to cause diarrhoea and vomiting in all age groups and mortality in preweaned pigs. Mortality rates are reported to be lower than in outbreaks of PED. SDCV is not a zoonosis or a risk to other animals. There are numerous coronaviruses that can cause infections in animals, and the enhanced surveillance and testing currently ongoing in North America is likely to have increased the likelihood of finding additional coronavirus in pigs.
Clinical cases due to this deltacoronavirus have also been made reportable in the US. The association of this SDCV with epidemic diarrhoea and its emergence in North America makes it important to develop the capability to detect the virus in pigs and allow prompt diagnosis of suspect cases, alongside diagnostic testing for other pathogens.
Further Reading
You can view the full report by clicking here.
Find out more about the diseases mentioned by clicking here.
June 2014






