



Study shows PRRS vaccine can provide protection against challenge with HP-PRRSV
Immune response, IL-10 and protective efficacy against a single HP-PRRSV challenge or in conjunction with PRRSV1 of pigs intradermally and intramuscularly vaccinated with modified live PRRSV1.Introduction
Co-existence of PRRSV1 and PRRSV2 has been increasingly reported in several Asian countries, including China, Korea, Vietnam and Thailand1. In the presence of co-infection with both PRRSV genotypes, clinical diseases are more severe compared to a single infection with either genotype. The study was conducted to test the efficacy of UNISTRAIN® PRRS when administered intramuscularly or intradermally in pigs against a challenge with a PRRSV2 and a challenge with co-infection with PRRSV2 and PRRSV1.
Materials and Methods
Forty-two PRRSV free pigs at 3 weeks of age were randomly allocated into 7 groups of 6 pigs each. Groups VacIM/PRRS2 and VacIM/PRRS1+2 were intramuscularly vaccinated with UNISTRAIN® PRRS. Groups VacID/PRRS2 and VacID/PRRS1+2 were intradermally vaccinated with UNISTRAIN® PRRS. Group NV/PRRS2, NV/PRRS1+2 and NV/Unch were vaccinated with PLACEBO. At 35 days post vaccination, groups VacIM/PRRS2, VacID/PRRS2 and NV/PRRS2 were intranasally challenged with FDT10US23 isolate (HP-PRRSV) and groups VacIM/PRRS1+2, VacID/PRRS1+2 and NV/PRRS1+2 were intranasally challenged with a co-infection of AN06EU4204 and FDT10US23 isolates (PRRSV1 and HP-PRRSV, respectively), at 1 ml of each isolate/nostril. Group NV/Unch was kept as non-vaccinated/non-challenge control. Following vaccination and challenge, pigs were monitored for PRRSV quantification by RT-qPCR, safety of the vaccine by IL-10, immune response including ELISA and IFN-γ-PC and macroscopic lung lesions.
Results
Following vaccination, ID vaccinated pigs had shorter viraemic phase and lower RNA level compared to IM vaccinated pigs. ID vaccinated pigs had significantly lower IL-10 level than IM vaccinated pigs (Figure1), but IFN-γ-PC were significantly higher (Figure2). There was no difference in antibody response.
No macroscopic lung lesions were observed in pigs of the NV/Unch group throughout the experiment. In contrast, pigs in the NV/PRRS2 and NV/PRRS1+2 groups had significantly higher scores compared to that of the vaccinated challenged groups at 7 DPC. The NV/PRRS1+2 groups had the significantly highest score. Macroscopic lung lesions in the VacID/PRRS2 and VacID/PRRS1+2 groups were significantly lower compared to that of the VacIM/PRRS2 and VacIM/PRRS1+2 groups.
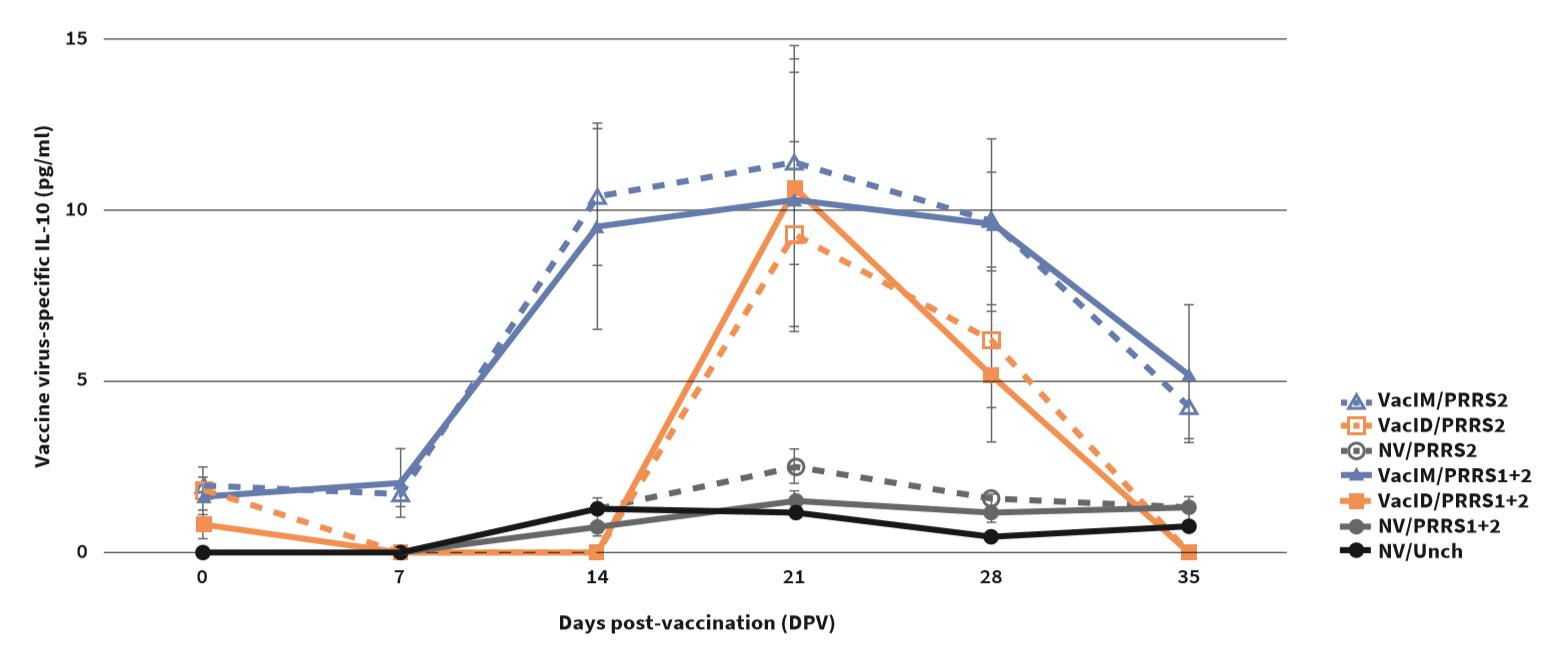
Porcine interleukin-10 (IL-10) concentration in cell culture supernatants of stimulated PBMC with vaccine virus. Values are expressed as mean±SEM. Different letters indicate statistical (P<0.05) differences among groups.
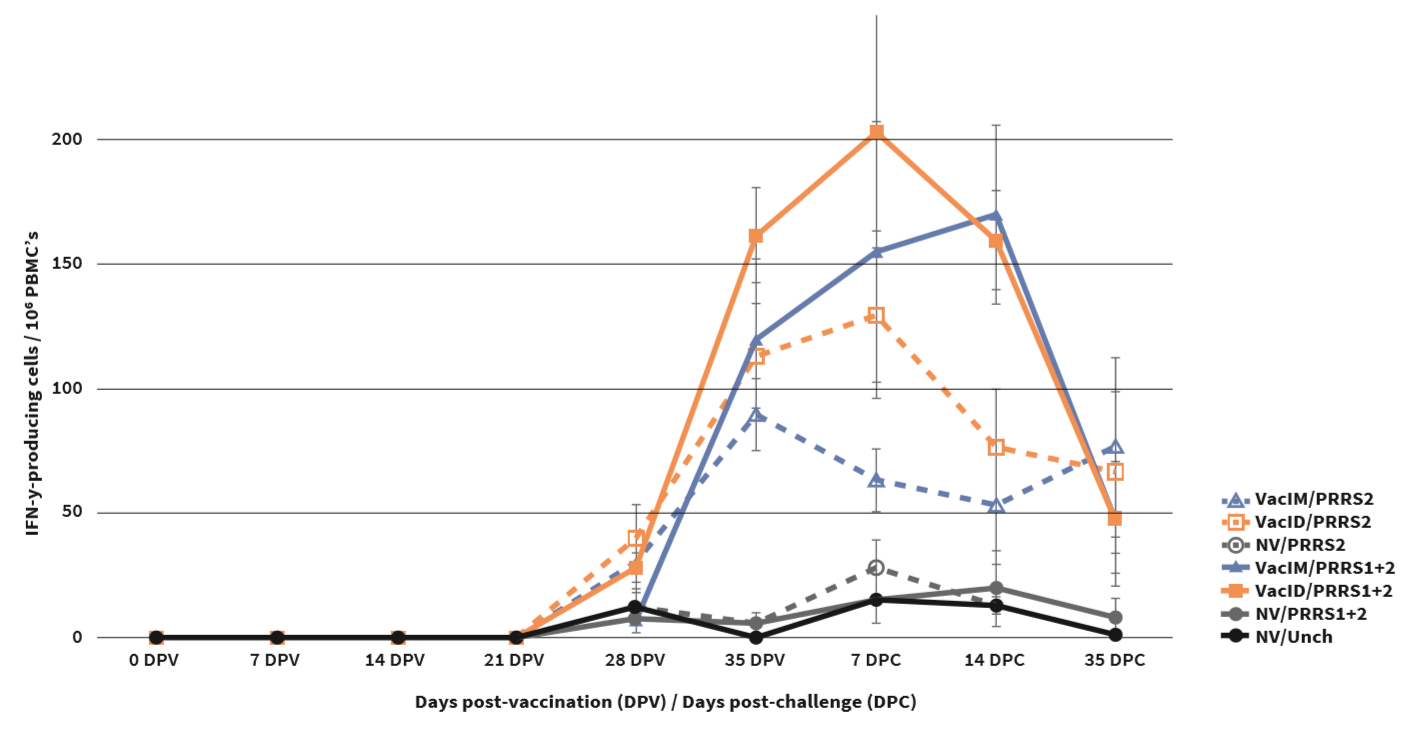
PRRSV-specific IFN-γ-producing cells (IFN-γ-PC) in stimulated PBMC with: (A) homologous virus; (B) heterologous type 1 PRRSV (AN06EU4204); and (C) heterologous type 2 PRRSV (FDT10US23). Values are expressed as mean±SEM. Different letters indicate statistical (P<0.05) differences among groups.
DISCUSSION In conclusion, the results of the study suggested UNISTRAIN® PRRS administered, either by ID or IM, can provided protection against challenge with HP-PRRSV, either alone or in conjunction with PRRSV1 as demonstrated by reduced lung lesion and viremia. ID route might represent an alternative to improve vaccine efficacy as it provided lower IL-10 and higher IFN-γ-PC.
| References | ||||
|---|---|---|---|---|
| 1. Nilubol et al. | ||||
| (2013) | . Arch Virol | 158(5):943-53 |








