



African swine fever in wild boar: proper disposal of offal
Offal of ASF-infected wild boar is the source of the ASF virusEditor's note: the following is an excerpt from African swine fever in wild boar populations - ecology and biosecurity. It was created by the FAO, WOAH and European Commission. Additional content from the booklet will be shared as an article series.
The offal of ASF-infected wild boar is the source of the ASFV and, if not handled with appropriate biosecurity measures can be a source of virus spread.
All animal parts must be removed from the forest. The easiest method is to bury parts in a designated pit that must be approved by the environmental protection authority or veterinary service. The pit should be close to the dressing area and directly excavated in the ground with consideration given to the groundwater level. The size of the pit must be able to contain the expected amount of offal per hunting season and must also be deep enough to prevent the scavenging of other wild animals (including boar). The pit area should be fenced and have a lockable gate. This method of offal disposal is practical wherever digging is possible.
When completely full, a pit can be closed and a new one excavated. Alternatively, and where allowed, its contents can be removed and safely disposed under the supervision of the veterinary service. Containers are a valid alternative to pits. Usually sealable and leak-proof plastic containers (500–600 litres in size) are placed close to dressing areas and then emptied when needed following the instructions provided by the veterinary service. Reused pits or containers are of evident advantage when rendering plants accept animal waste and offal.

SAFE ON-SITE STORAGE OF HUNTED WILD BOAR UNTIL AN AFRICAN SWINE FEVER-NEGATIVE TEST RESULT
In ASF-infected areas, hunted wild boar cannot leave the hunting ground without an ASF-negative test result. Official veterinary laboratories must carry out ASF tests. The results obtained by commercial kits that are available on the market in some countries are completely unreliable and their use is inappropriate for the eradication of the infection.
Each hunting ground should be equipped with refrigerators in which, after dressing and sampling, the entire wild boar is stored and individually identified. If the carcass is divided into several pieces (this is not recommended), each piece must be clearly identified, with the number of pieces obtained from a single wild boar registered. No part of the animal (including trophies) can leave the hunting ground before the hunted wild boar has an ASF-negative test result.
Storage and sample activities must be organized to avoid the release of ASF-negative animals while test results are still pending for other individuals. Animals should be stored as batches and released only when each entire batch tests negative for ASF. The procedure is easy to manage when hunting is carried out exclusively during weekends and requires more careful planning for different timings (hunting, sampling, testing and release of ASF-negative animals).
Cold storage facilities or refrigerators for hunted wild boar carcasses can be installed in closed dressing facilities or in a hunting lodge and must be cleaned after the removal of carcasses or meat.
In Poland, the veterinary service provided transportable storage rooms for wild boar. The carcasses can be dressed outside these room, with offal collected in containers and animals stored until laboratory results are received.


PROCEDURES FOR THE DISPOSAL OF WILD BOAR WITH AFRICAN SWINE FEVER AND FOR CLEANING AND DISINFECTING FACILITIES
In the event of a positive ASF result, all the stored carcasses (or pieces of meat) must be safely disposed of by the veterinary service. The dressing area, cold storage facilities or refrigerator must also be cleaned and disinfected. Since the inactivation of the virus in the dressing area, in refrigerators and from clothes, vehicles and tools is based on cleaning and disinfection, hunters should be trained and provided with written instructions. Preliminary cleansing is needed before the use of any disinfectants. Mechanical brushing with a detergent solution is highly effective in cleaning contaminated surfaces and objects and is important for disinfection to be effective. Only freshly prepared disinfectant solutions should be used and for the required time necessary to be effective (up to 60 minutes contact time).

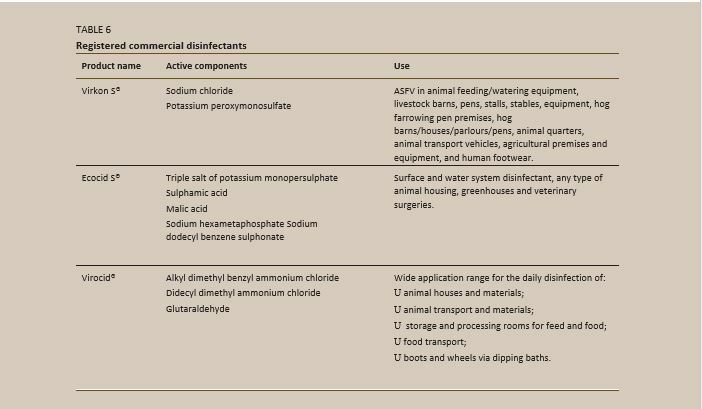
DISINFECTANTS RECOMMENDED FOR AFRICAN SWINE FEVER VIRUS
- chlorine (sodium hypochlorite)
- iodine (potassium tetraglicine triiodide)
- quaternary ammonium compound (dodecyl dimethyl ammonium chloride)
- vapo-phase hydrogen peroxide
- aldehydes (formaldehyde); organic acids
- oxidizing acids (peracetic acid)
- alkalis (calcium hydroxide and sodium hydroxide)
- ether and chloroform



KEY MESSAGES
- In the infected areas, each hunting ground must develop a simple, basic, biosecurity management plan. The main goal is to prevent the viral contamination of the environment and the mechanical transportation of the virus outside the hunting ground through hunting and related activities.
- Each hunting ground must organize a wild boar dressing area and offal and wild boar storage facilities.
- Hunted wild boar should be individually identified and safely stored in the hunting ground until tested negative for ASF.
- If a hunted wild boar tests positive for ASF, all the stored animals, regardless of the species, must be disposed of under the supervision of the veterinary service.
- Hunting must be reauthorized when cleansing and disinfection of the infected hunting ground facilities are completed.
Guberti, V., Khomenko, S., Masiulis, M. & Kerba S. 2022. Chapter 4. African swine fever in wild boar – Ecology and biosecurity. Second edition. FAO Animal Production and Health Manual No. 28. Rome, FAO, World Organisation for Animal Health and European Commission. https://doi.org/10.4060/cc0785












